Recently, our company's invasive brain-computer interface (BCI) stroke rehabilitation system project successfully completed animal trials, obtaining complete and effective core experimental data. This milestone lays a solid foundation for subsequent technological optimization and clinical research.
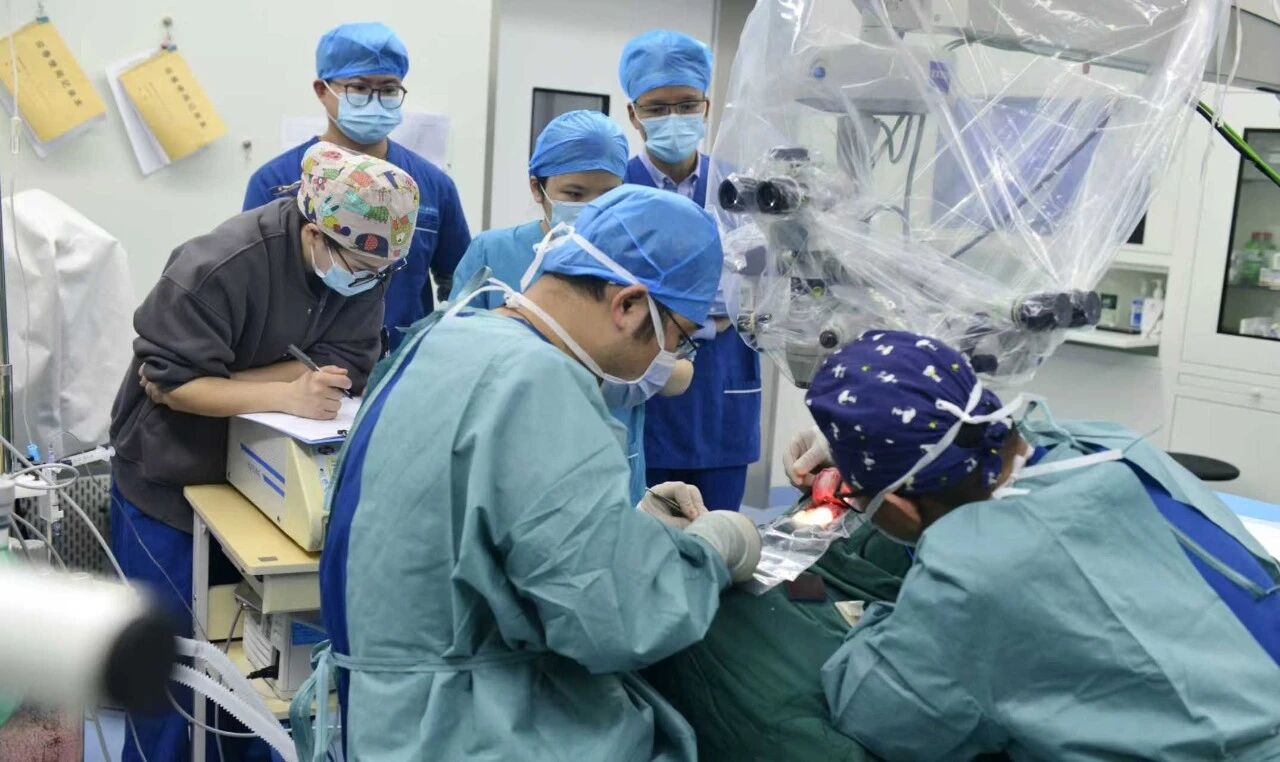
Post-stroke limb motor dysfunction affects hundreds of millions of patients worldwide, presenting a persistent challenge that traditional rehabilitation methods struggle to overcome due to their limited capacity to trigger deep neural remodeling. Addressing this urgent clinical need, our company has developed a proprietary invasive BCI rehabilitation system. Leveraging precision neural signal decoding technology, the system captures motor intention code from the patient's brain, forming a closed-loop training circuit with external rehabilitation devices. Through technological innovation, it activates neuroplasticity, helping patients rebuild motor function and effectively improve their quality of life.
As a core procedure in the implementation of high-risk medical devices such as invasive BCIs, animal trials serve multiple critical functions: scientific validation, risk assessment, and technological iteration. They represent a mandatory requirement under China's Class III medical device regulations and serve as an essential bridge between laboratory research and clinical application. These trials strictly adhered to ethical principles for animal research, comprehensively validating product performance in vivo models. They successfully demonstrated the feasibility of stroke animal models, the electrophysiological mechanisms of invasive rehabilitation therapy, stimulation pulse waveform frequencies, precise threshold settings and stimulation safety protocols, and the establishment of closed-loop control and feedback mechanisms.
With 28 years of dedicated research in the BCI field, IntelliMicro has consistently remained at the forefront of technological innovation, accumulating substantial technical expertise and extensive R&D experience. Currently, our entire team is focused on deepening the integration of medical requirements and engineering technology, accelerating the transition from technological achievement to industrial application—the critical journey "from 1 to 100"—with the ambition of defining and leading the clinical transformation of BCI technology.
The successful completion of these animal trials represents a pivotal advancement in IntelliMicro's BCI project implementation. Moving forward, our company will maintain unwavering determination and full confidence, leveraging our core strengths to accelerate the iterative upgrade and clinical deployment of the stroke rehabilitation system. True to our commitment to universal healthcare, we will transform medical technology into accessible, reliable, and affordable treatment solutions, safeguarding patient health worldwide with IntelliMicro's capabilities and contributing powerful momentum to the construction of a global health community.


